A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about Lewis acids is true?
A) Lewis acids are proton donors.
B) Lewis acids are proton acceptors.
C) Lewis acids are electron pair donors.
D) Lewis acids are electron pair acceptors.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the strongest base?
A) CH3COCH3
B) CH3COOH
C) NH3
D) H2O
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the direction of equilibrium when acetylene (C2H2) reacts with H2N- in an acid-base reaction? 
A) Left
B) Right
C) Neither
D) Cannot be determined
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is not a Lewis acid?
A) AlCl3
B) HCl
C) H2O
D) CBr4
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electrophilic site in the following compounds? 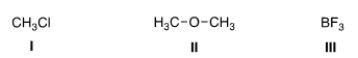
A) I = Carbon; II = carbon; III = boron.
B) I = Chlorine; II = carbon; III = boron.
C) I = Carbon; II = oxygen; III = boron.
D) I = Carbon; II = carbon; III = fluorine.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is the strongest acid?
A) CH3OH
B) BrCH2OH
C) CH3NH2
D) CH3Cl
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements explain why HBr is a stronger acid than HF?
A) Br- is more stable than F- because Br- is larger than F-.
B) Br- is less stable than F- because Br- is larger than F-.
C) Br- is more stable than F- because Br- is less electronegative than F-.
D) Br- is less stable than F- because Br- is less electronegative than F-.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 48 of 48
Related Exams