A) 1
B) 2
C) 3
D) 4
E) 8
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonia's unusually high melting point is the result of
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonding.
E) ionic bonding.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A temperature increase causes __________________ in the conductivity of a conductor.
A) a decrease
B) an increase
C) an increase or decrease (depending on the conductor)
D) a modulation
E) no change
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What word best describes the type of liquid crystal represented below? 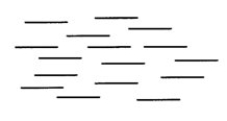
A) nematic
B) cholesteric
C) smectic
D) isotropic
E) elastic
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following phase diagram and identify the feature represented by point B. 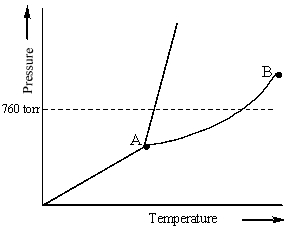
A) melting point
B) triple point
C) critical point
D) sublimation point
E) boiling point
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following liquids is likely to have the highest surface tension?
A) Br2
B) C8H18
C) CH3OCH3
D) CH3OH
E) Pb
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 5.00 g sample of water vapor, initially at 155°C is cooled at atmospheric pressure, producing ice at -55°C. Calculate the amount of heat energy lost by the water sample in this process, in kJ. Use the following data: specific heat capacity of ice is 2.09 J/gK; specific heat capacity of liquid water is 4.18 J/gK; specific heat capacity of water vapor is 1.84 J/gK; heat of fusion of ice is 336 J/g; heat of vaporization of water is 2260 J/g.
A) 15.6 kJ
B) 10.2 kJ
C) 5.4 kJ
D) 3.2 kJ
E) 1.6 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The meniscus of mercury in a glass capillary tube is convex because of
A) the very high density of mercury as compared with water.
B) the low surface tension of mercury.
C) the greater attraction of mercury atoms to the glass than to each other.
D) the weaker attraction of mercury atoms to the glass than to each other.
E) electrostatic repulsion between the glass and the mercury.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the adhesive forces between a liquid and the walls of a capillary tube are greater than the cohesive forces within the liquid
A) the liquid level in a capillary tube will rise above the surrounding liquid and the surface in the capillary tube will have a convex meniscus.
B) the liquid level in a capillary tube will rise above the surrounding liquid and the surface in the capillary tube will have a concave meniscus.
C) the liquid level in a capillary tube will drop below the surrounding liquid and the surface in the capillary tube will have a convex meniscus.
D) the liquid level in a capillary tube will drop below the surrounding liquid and the surface in the capillary tube will have a concave meniscus.
E) None of the above will occur.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In a transistor, the current through one semiconductor junction controls the current through a neighboring junction.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Smectic liquid crystals are more highly ordered than either nematic or cholesteric liquid crystals.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 101 - 111 of 111
Related Exams