A) [Ar]4s24d4.
B) [Ar]4s24p63d3.
C) [Ar]4s23d9.
D) [Ar]3d9.
E) [Ar]4s13d10.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? [Kr]5s24d105p3
A) Sn
B) Sb
C) Pb
D) Bi
E) Te
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
When l = 1 then ml can be ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List the following types of radiation from lowest frequency to highest frequency: microwave, X ray, ultraviolet, visible, and infrared
A) microwave < infrared < visible < ultraviolet < X ray
B) X ray < ultraviolet < visible < infrared < microwave
C) visible < ultraviolet < microwave < X ray < infrared
D) infrared < X ray < microwave < ultraviolet < visible
E) infrared < visible < microwave < ultraviolet < X ray
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
For all atoms of the same element, the 2s orbital is larger than the 1s orbital.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
"No two electrons in an atom can have the same four quantum numbers" is a statement of
A) the Pauli exclusion principle.
B) Bohr's equation.
C) Hund's rule.
D) de Broglie's relation.
E) Dalton's atomic theory.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a correct set of quantum numbers for an electron in a 5f orbital?
A) n = 5, l = 3, ml = +1
B) n = 5, l = 2, ml = +3
C) n = 4, l = 3, ml = 0
D) n = 4, l = 2, ml = +1
E) n = 5, l = 4, ml = 3.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct electron configuration for gold?
A) [Xe]4f145d96s2
B) [Xe]4f145d106s1
C) [Xe]4f135d106s2
D) [Xe]4f145d105s1
E) None of the electron configurations is correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
________ is the element represented by the electron configuration [Ne]3s2 3p1.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which scientist proposed that energy of radiation is composed of extremely small indivisible packages called quanta? ("Quanta" is the plural of "quantum.")
A) Max Planck
B) Louis de Broglie
C) Clinton Davisson
D) Sir Isaac Newton
E) Lothan Meyer
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Pauli exclusion principle states that no ________ electrons within an atom can have the same ________ quantum numbers.
A) 4; 6
B) 2; 4
C) 3; 6
D) 6; 10
E) 3; 8
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The orientation in space of an atomic orbital is associated with
A) the principal quantum number (n) .
B) the angular momentum quantum number (l) .
C) the magnetic quantum number (ml) .
D) the spin quantum number (ms) .
E) none of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct electron configuration for a germanium (Ge) atom?
A) 1s22s22p63s23p64s24p2
B) 1s22s22p63s23p64s23d104p2
C) 1s22s22p63s23p2
D) 1s22s23s23p5
E) None of the answers is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the arrangement of electromagnetic radiation which starts with the lowest energy and increases to the greatest energy.
A) radio, infrared, ultraviolet, gamma rays
B) radio, ultraviolet, infrared, gamma rays
C) gamma rays, infrared, radio, ultraviolet
D) gamma rays, ultraviolet, infrared, radio
E) infrared, ultraviolet, radio, gamma rays
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are there in the 2nd principal energy level (n = 2) of a phosphorus atom?
A) 3
B) 5
C) 6
D) 8
E) 10
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The unusual electron configuration of Mo indicates the
A) unusual stability of half-filled d-orbitals.
B) unpredictability of the electron filling patterns of the non-representative elements.
C) unusually large size of Mo atoms.
D) greater than expected electronegativity of Mo.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The FM station KDUL broadcasts music at 99.1 MHz. Find the wavelength of these waves. (c = 3.00 × 108 m/s)
A) 1.88 × 10-2 m
B) 0.330 m
C) 3.03 m
D) 5.33 × 102 m
E) > 103 m
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following sets of quantum numbers can correctly represent a 3p orbital? 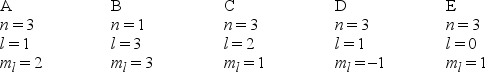
A) A
B) B
C) C
D) D
E) E
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The electron configuration for chlorine is [Ne]3s2 3p5.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Emission spectra
A) cannot be used to identify an unknown atom.
B) can be used to identify unknown atoms.
C) can be explained by the movement of protons colliding with electrons.
D) None of these choices.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 135
Related Exams